Image in header: Redox cartoon. Source: https://www.youtube.com/watch?v=_HZCMuw2BG4 (Jwalpa Coaching Classes)
To turn dead matter into living things, you need metabolism. In 0.3. we saw that CHNOPS are the elements that dominate in the construction and functioning of living cells (carbon, hydrogen, nitrogen, oxygen, phosphorus, sulphur). All these elements have to be taken up by living beings from the environment , and built into the (cell) body in the form of biomolecules. However, the most central element is carbon. To understand the basics of metabolism, we are going to focus on this central element.
Oxidation and reduction of carbon
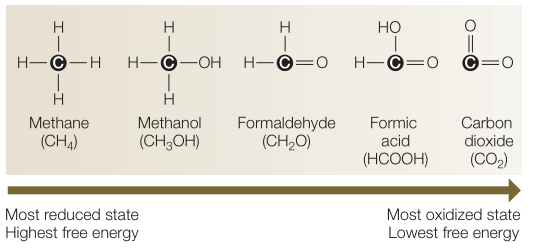
The carbon most commonly found in our Solar System (in elsewhere in the universe) is the oxidised formCO2 or CO. If life wants to use this carbon to build the cell body, then that carbon has to be reduced. What do we mean by ‘reduced’ and ‘oxidised’ carbon?
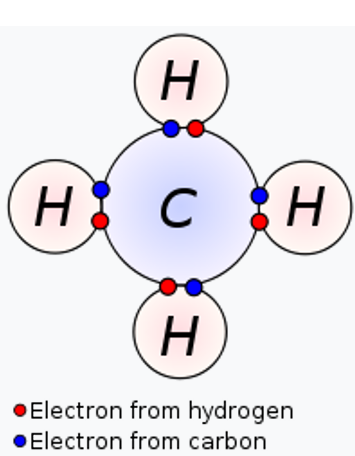
Carbon is an atom with four electrons in the outer ‘shell’. Every atom prefers to have eight in the outer shell, because that is the most stable state. This is why carbon likes to form chemical bonds to share four electrons from other atoms. InCO2, for example, carbon is going to give two electrons to the bond with oxygen on both sides itself, and oxygen itself also shares two. that way, you arrive at a total of 8 electrons.

Because in the bond between oxygen and carbon, the two partner atoms are not the same, there is always one that attracts those shared electrons more to itself. Oxygen is more ‘electronegative’ than carbon. That means oxygen pulls the electrons of the molecule (the shared ones) more towards itself. So in this C-O compound, you can say that carbon loses electrons to oxygen. A carbon atom that has lost electrons is called ‘oxidised’. The electronegativity of carbon is 2.55 and that of oxygen is 3.44.
Now compare that to a carbon atom contained in the CH4 molecule.
Hydrogen has an electronegativity of 2.20. This is therefore less than carbon (2.55). In this C-H compound, this time carbon will attract the shared electrons to itself. So in this case, carbon has gained (rather than lost) electrons, and is thus ‘reduced’. So in this molecule, the carbon has attracted more negative charge to itself (more negative is a reduction).
As a rule, if you want to reduce oxidised carbon, you have to add energy. Whereas oxidising already reduced carbon adds energy. This is exactly what life makes use of. As a rule (at least on Earth), life is going to use energy from the environment to start reducing oxidised carbon. Then that life is going to oxidise that reduced carbon back and store the resulting energy to use for all kinds of its own life processes. The best-known terrestrial example of this is photosynthesis. Life uses solar energy to reduce the carbon ofCO2 from the air, turning it into, for example, glucose (C6H12O6). Then some of that glucose is consumed back: the carbon is oxidised back thus. The oxidation of those glucose carbons releases energy, which a living cell will harvest and store in the ATP molecule. That energy molecule is later used for just about anything a living cell wants to do.
Redox reactions as basis for life
The Nobel laureate Albert Szent-Györgyi (a biochemist) once said, “Life is nothing but an electron looking for a place to rest”. By this he meant that life is based on that change from oxidised carbon to reduced carbon, and back again. The electron has then ‘found rest’ when it has gone back off the carbon and gone back to oxygen, for example. ‘Redox’ is the abbreviation for Reduction Oxidation. Every form of life has this as the most essential activity in its metabolism, and derives the necessary ‘life energy’ from it.
To perform the reduction part of the redox reactions, an organism has to find an electron somewhere to give off to the carbon. The molecule used for this can be anything, and is called the electron donor. When oxidising carbon, the organism will want to donate the electron to an electron acceptor. Different molecules can be used for this too. We will come across several examples of this later.