
Image in header: The glucose molecule is a universal source of chemical energy for all living cells on Earth, whether with or without oxygen. Source: https://www.instagram.com/p/Cx7u7R5RkDm/ (@Microbiologyupdate).
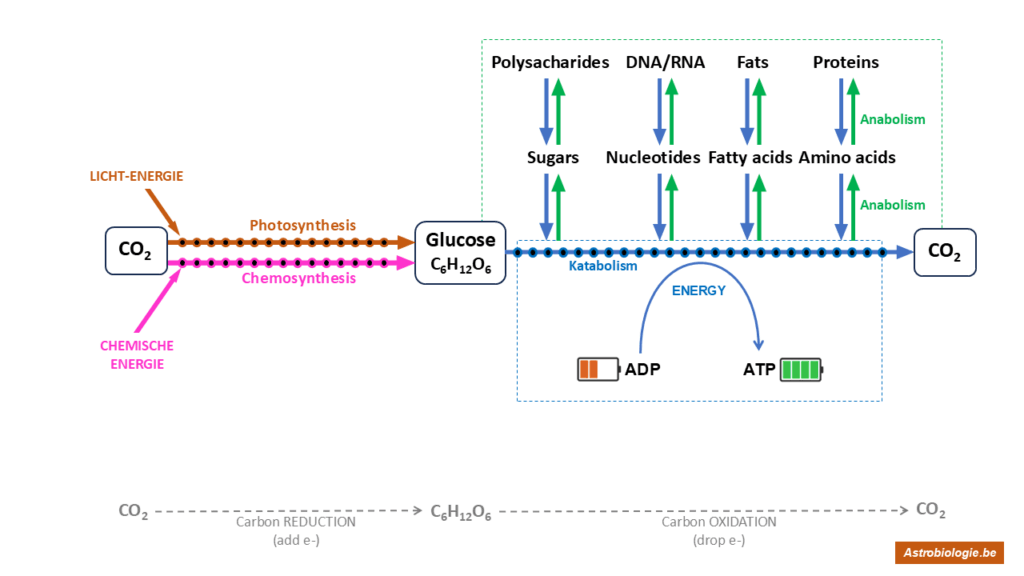
On a planet where there is life, the carbon ofCO2 is massively reduced by that life. On the one hand, to make biomolecules from which the living cell builds its body. On the other hand, to break down (oxidise back) that molecule of reduced carbon and thus harvest energy from it. the diagram below summarises this. We will come back to that diagram further.
CO2 is found everywhere in the Solar System (and beyond) in large quantities. It is very common. It is also reduced without life (abiotically) in spontaneous reactions, but not as massively as by life. The spontaneously reduced carbon occurs in a wide variety of molecules, but not in large more complex organic molecules, not in large ‘biomolecules’ in other words. Many organic molecules have already been discovered on asteroids and comets. One of the most frapante recent discoveries comes from the sample brought to Earth by the OSIRIS-REX mission (NASA) from the asteroid Bennu. in this sample were found all five nucleotides from which DNA and RNA are made, as well as 14 of the 20 amino acids that terrestrial life uses to build all its proteins. So it may be clear that carbon can be spontaneously reduced without the intervention of life, and that even most of the basic units from which biomolecules are made can indeed arise by themselves. But again: massively reducing carbon to then make equally massively more complex organics with it, only life does that as far as we know.


Now let’s return for a moment to the diagram that gives an overview of all metabolism. It shows how living cells use the common oxidised form of carbon –CO2 – to build up their own bodies on the one hand, and to harvest energy from the reduced form of carbon on the other. GLUCOSE is the molecule symbolised in the diagram for the reduced form of carbon. But how can this simple molecule stand for a super-complex fact like living cells with all their chemical activity? Life is made up of four major groups of biomolecules (fats, sugars, proteins, nucleic acids), not just glucose. How about that?
The very comprehensive and complete answer can be found on Roche Biochemical Pathways’ world-famous poster. Fans who do not yet have this poster at home should order it as soon as possible using the button below. They will deliver it by post for free! You can also already download its digital image here on astrobiology.co.uk.
It is an overview of all the essential biochemical steps that living cells use to turn glucose or other food into all the biomolecules they need. By following certain pathways shown on the poster, you can basically make any fats, proteins, nucleic acids or sugars you want. And living cells are built in such a way that they have all the enzymes and structures to go through those particular biochemical pathways (‘pathways’). The opposite direction is also possible. If we eat other organisms, we can convert all those eaten biomolecules back into glucose or into other biomolecules in our cells.
Why was glucose chosen as the starting point for every biochemical pathway needed? Well, glucose is effectively the start of the most commonly used pathways (‘pathways’) through life on Earth. In this course, very succinctly summarised anyway, we are going to take a look at which pathways those are mainly. Further on in this ‘chapter ‘part 2’ we will discuss several examples.
Autotrophs are the ‘producers’
We can divide the diagram below into left and right halves. Not all life can complete the diagram from left to right.

For the left half: to convertCO2 (oxidised carbon) into glucose (reduced carbon), you have to belong to the ‘autotrophic’ organisms. Autotrophic literally means self-feeding. These organisms are capable of self-reducingCO2 from the air or water, i.e. adding an electron to the carbon. After many intermediate steps, the starting molecule glucose is created, which all life can continue with as a staple food. We also call this process ‘carbon fixation’. There are a total of 6 known ways to do carbon fixation. All 6 use energy from the environment and grab an electron of another molecule from the environment to enable the reduction of carbon. That molecule from which they grab an electron is called the electron donor. Some use light energy for this process (photosynthesis), while others use binding energy from molecules in the environment (chemosynthesis). We are going to discuss examples of both under ‘milestones’ of life on Earth. The organisms that can do carbon fixation thus provide food for everyone: for themselves and for all heterotrophic (= non-autotrophic) organisms. We therefore also call them the ‘producers’.
Everyone is a ‘consumer’
Autotrophs are thus able to fix carbon (the left: fromCO2 to glucose). Heterotrophs cannot do this, and thus must always feed on biomolecules once made by autotrophs. Heterotrophs are only on the right side of the diagram, where biomolecules are made from glucose on the one hand (fats, DNA/RNA, sugars and proteins) and glucose is broken down to harvest energy from it on the other. That energy is always stored in ATP molecules (see below). The biomolecules to build up the cell body are made from all kinds of intermediates that uti the breakdown of glucose. The building up of new biomolecules is called anabolism (the ‘building up’ part of metabolism). Because heterotrophs use the food originally made by autotrophs, they are called consumers.
But make no mistake, the autotrophs are, of course, consumers themselves. They too are on the right side of the diagram. Every living cell has to build its body by making all kinds of biomolecules, and every living cell constantly needs energy (ATP) to stay alive and do everything a cell does. In other words, all living things on Earth are consumers, and only some are also producers.
Recall that the autotrophs borrowed an electron from some external molecule (the electron donor) to reduce carbon. You will see from the examples later that at the end of the ride, after the breakdown of glucose (catabolism), an electron is eventually given back to another molecule, the electron acceptor. The best-known example of such an electron acceptor is oxygen, the reason why we breathe. Once the electron has been given to oxygen, it is going to be converted intoH2O, i.e. water.
The Earth today with plants and animals
On Earth, life is dominated by two variants of the principles described above. Carbon fixation occurs mainly through photosynthesis in which water is the electron donor, emitting oxygen gas. This is the work of the dominant producers, namely all plants and Cyanobacteria. In turn, the dominant consumers are all organisms that eat other organisms (heterotrophs) and engage in respiration with oxygen gas (aerobic respiration). Oxygen is the electron-acceptor here. All complex life forms do this: plants, animals, fungi, and complex unicellular organisms. There are also a lot of Bacteria and Archaea that also do this.

That these forms of metabolism are dominant in terrestrial life has not always been the case. And throughout Earth’s history, other forms of metabolism have also persisted and been important in the whole cycle of nutrient and energy metabolism. Even today, this is still true. For astrobiologists, it is crucial to understand all these various forms of metabolism on the ground. We need it to understand the origin of life on a planet, and to estimate which places are habitable or not. A lot of organisms live in extreme places where alternative forms of metabolism are required to survive. We certainly cannot limit ourselves to conditions like those on today’s planet Earth, where sunlight and oxygen are abundantly available.
