
Image in header: Cyanobacteria are the inventors of oxygenic photosynthesis on Earth, splitting water and emitting oxygen gas. Here we see cells of the genus Prochlorococcus, which is responsible for 20% of our planet’s current oxygen production. Source: https://www.planetary.org/space-images/cyanobacteria-under-the-microscope
Oxygen gas (O2) is a molecule with distinct properties. For living things, it is actually a dangerous molecule because it can be so reactive (be sure to read Nick Lane’s book ‘Oxygen’ to understand more about this). On the other hand, on Earth it has enabled a form of life that can extract extra energy from its metabolism: aerobic cellular respiration. Chapter 2 will elaborate on the different forms of metabolism on Earth. We will then see that the presence of oxygen in the atmosphere and in the oceans has played a crucial role in the history of life and made possible the present earthly paradise with that enormous biodiversity. But our planet took billions of years to do so.
Acidoph gas is very abundant in our atmosphere today thanks to its production by living things engaged in photosynthesis. The current level of 21% is maintained because green bacteria and plants constantly produce new oxygen. But in the first 4 billion years of Earth’s history, oxygen levels were much lower. Heinrich Holland in 2006 made a survey of Earth’s oxygen concentrations in five major phases, as shown on the figure below. Let us briefly review these phases.
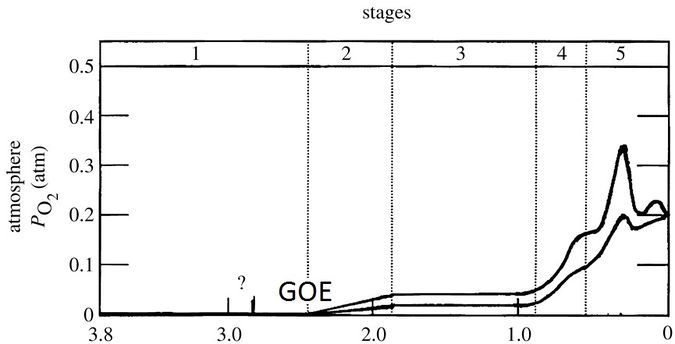
Estimate of atmospheric oxygen concentrations on Earth. The numbers on the X-axis indicate time (0 = today, 3.8 = 3.8 billion years ago). On the Y axis, we see the partial oxygen pressure (e.g. 0.1 corresponds to 10% of the air consists of oxygen gas). A lower limit and upper limit are indicated. These correspond to minimum and maximum estimates of oxygen pressure in that perdiode. GOE = Great Oxygenation Event. Source: “The oxygenation of the atmosphere and oceans” Heinrich Holland, The Royal Society Publishing Philosophical Transactions B, June 2006.
Phase 1: the first 2 billion years: little oxygen production and rapid consumption
When our planet formed, there was a very intense period of ‘outgassing’, as discussed earlier. Water vapour was massively present in that early atmosphere. Back then, there was no protective ozone layer (because you need a lot of free oxygen for that), so the sun’s high-energy UV radiation could bombard those water molecules in the air unhindered. As a result, water high in the atmosphere started splitting into oxygen gas and hydrogen gas, without the intervention of living cells. The hydrogen gas is very light, and escapes easily into empty space. As a result, the Earth’s atmosphere became slightly enriched with oxygen gas. The production of oxygen gas in this way is also seen elsewhere in space. The Belgian instrument ROSINA, for example, was able to measure up to 10% free oxygen around comet 67P Churyumov-Gerasimenko in 2015. It is rather obvious that there are no photosynthetic organisms living on that comet. Anyway, the production of oxygen in this way is negligible in quantity compared to the subsequent poduction of oxygen on Earth by photosynthesis. This oxygen was immediately captured again by all kinds of molecules in the atmosphere (other volcanic gases), on land and in the oceans. We are talking about the turbulent and unstable period of the Hadean Era, from 4.54 to 3.9 billion years ago.
But even in the subsequent period, more specifically up to 2.4 billion years ago, we find that the Earth’s atmosphere and oceans remained virtually oxygen-free. Atmospheric concentrations are estimated at less than 2 ppm, which is actually virtually nothing. Rocks from this period contain almost no oxidised minerals. Studies of sulphur isotopes from that time also show that the atmosphere was quasi-oxygen-free. The Earth was certainly populated with single-celled life by then. Could life then have waited so long to ‘invent’ photosynthesis?
No. All indications are that photosynthesis occurred relatively quickly from the time living cells appeared in the light-rich layers of the oceans. The first forms of photosynthesis were anoxygenic, though. This means that the emission from those early photosynthetic cells was not oxygen gas. Anoxygenic photosynthesis still exists today in some bacterial species. More on this in the great chapter on the characteristics of life. The next step was oxygenic photosynthesis: cells that can use sunlight to split water molecules for their metabolism, and emit oxygen as a waste product. We are talking about the group of cyanobacteria. The strongest evidence that this oxygenic form also rapidly emerged is found in the so-called banded iron formations (BIFs).
BIFs are clearly delineated layers in fossil ocean floors that are full of rusted iron. So these are iron oxides in various forms, where the iron atom has a charge of +3, noted as Fe3+. However, the iron released from volcanic processes (on the seabed or on land) is mainly iron in reduced form, or Fe2+. This reduced iron thus has one more electron, and is therefore an easy ‘victim’ for the reactive oxygen molecule, which is always looking for electron-rich atoms to bind to. As it were, an oxygen atom is always hungry for an electron it can grab from another atom somewhere. Oxidising actually means giving up an electron by sharing it with an electron-hungry atom like oxygen.
The early oceans must have been well filled with reduced iron (and other reduced molecules). In such environments, newly created oxygen will immediately be captured by those reduced substances. Now, reduced iron dissolves very easily in seawater. But when it reacts with oxygen to form iron oxide, it no longer dissolves. The iron oxides then fall down to the bottom. Et voilà! So these precipitated layers of iron oxides are the BIFs.

We find quite large amounts of BIFs in rocks from 3.9 to 2.4 billion years old. So it is clear that oxygen was added to the oceans several times during that period. This is easily explained by the rise of cyanobacteria with their oxygenic photosynthesis. An additional clue to this is the presence of chemical traces found in rocks of 2.7GA, namely chemical residues of photosystem 2 (PSII), the biochemical complex required by those bacteria to engage in oxygenic photosynthesis. However, we can see that total oxygen production always remained much smaller than the amount of reduced substances in the sea. In other words, oxygen was always captured in the ocean faster than it could be produced. So this leaves the atmosphere free of oxygen gas, despite the photosynthetic acitivity of the bacteria.
Phase 2: The biggest environmental disaster ever: the GOE
So for hundreds of millions of years, more oxygen was captured than was produced. By reduced iron, but also other common substances such as hydrogen gas, methane and sulphides. Even the decomposition of dead cells possibly consumed oxygen, but we don’t know that for sure at all. But the invention of oxygenic photosynthesis by cyanobacteria provided an undeniable advantage to these organisms, and eventually their populations would grow larger than our planet could even compensate for. What followed was a merciless impact on the entire planet, the so-called ‘Great Oxygenation Event’. An unprecedented environmental disaster actually.
The huge amount of photosynthetic bacteria started a very slow – but irreversible – addition of oxygen gas in our atmosphere (and also in the oceans). Many isotopic studies show that this increase lasted from 2.4 GA to 1.8 GA. A phenomenon that just kept increasing for 600 million years! Slowly but surely, then, the habitat changed globally and dramatically. And that change had historical consequences, such as:
- Increases in atmospheric oxygen concentrations of up to 10% of current concentrations
- The formation of BIFs was at its peak just before the GOE. But now the oceans everywhere are becoming too oxygen-rich, so reduced iron was already oxidised before it could spread to places where cyanobacteria grow luxuriantly. We see a decrease in BIFS as the GOE progresses. THE graph below shows an overview.
- Emergence of aerobic respiration: a form of metabolism that was more efficient than any previous one. Yet the production of oxygen by photosynthesis was even faster than the breakdown via cellular respiration. As a result, there was a surplus of oxygen that thus entered the atmosphere.
- An oxygen-rich environment that presumably stimulated the emergence of complex life (eukaryotes)
- The biggest extinction wave ever of organisms that dominated before, namely anaerobic life forms. These are cells we will discuss in the chapter on life, for which oxygen gas is nothing but dangerous poison. Such life forms do still occur today: in oxygen-deficient deep water layers, in swamps or in the intestines of humans and other animals, for example.
- Successive long ice ages, sometimes with ice sheets that occurred almost everywhere on the planet. The atmosphere previously contained a high concentration of the very potent greenhouse gas methane. This was oxidised to CO2 and water by the released oxygen. The CO2 is 28 times weaker in retaining heat, so the greenhouse effect reduced significantly. Water rained out quickly, especially when it got colder. At this time, there was also less solar energy than today. The main evidence for these intense ice ages consists of so-called diamictite (deposited by ice) in rocks 2.1 to 2.4 billion years old at various locations around the world.

Phase 3: Boring billion
Approximately between 1.8 billion years ago and 0.8 billion years ago (1.8GA-800MA) is a long period in Earth’s history with relatively few dramatic events. This period is therefore called ‘the boring billion’. The climate made few dramatic leaps, no super-sized milestones are known during this period for life, and geologically it was relatively ‘quiet’. This also means that there was fairly little new mountain formation, and therefore not much erosion. Thus, larger amounts of nutrients were also not entering the oceans. As a result, the existing populations of single-celled life also did not have a large conspicuous bloom. There were simple unicellular organisms (bacteria and Archaea), and presumably since the GOE also more complex cells: the Eukaryotes. Eukaryotic fossils are known to be over 1.6 billion years old, and biochemical traces were found that are even older and typical of eukaryotic cells. More on this in Part 2, where the milestones of life are discussed.

It seems somewhat strange that complex life (Eukaryotic) already existed in the ‘boring billion’, but that they waited more than a billion years to break through with their great biodiversity that we know today in every part of our planet. Multicellular complex life emerged and flourished only in the period after. The situation was as follows:
The shallow seas contained oxygen everywhere, the atmosphere too, about 1/10 of today’s oxygen concentration. The deeper layers, however, were virtually oxygen-free or anoxic. Due to the position of the continents and the relatively stable climate, there was hardly any exchange between the shallow and deep layers. Photosynthetic life needed light (only shallow), and the more complex eukaryotes needed oxygen (also only shallow). For them, the deep sea is unlivable. That deeper sea was a kind of disappearing-hole for essential nutrients and dead cells. Pretty little nutrition was already coming in because of limited geology (limited erosion of land). Moreover, there was little evolutionary pressure. If there are no major disruptions to the environment, e.g. from intense volcanism, climate fluctuations, or reversals in sea currents, then there is little pressure on existing life to innovate and thus give rise to new life forms.
Therefore, everything remained relatively stable for a very long time. Oxygen production by cyanobacteria continued quietly as before, without increasing, and the consumption of that oxygen by respiration also remained the same. So global oxygen levels did not change either. So what happened at the end of the ‘bore billion’ to get things moving again?
Phase 4: dramatic changes and snowball Earth
In the last 200-300 million years of the ‘boring billion’, a gigantic supercontinent named with Rodinia was formed. Most of this land was close to the equator. From about 800 million years ago (800 Ma), the mighty Rodinia began to fall apart again. As a result, this was a period of intense volcanism, as well as mountain formation and erosion. The rearrangement of sub-continents created new sea currents, also vertically between the deep and shallow ocean layers. Global climate made crazy leaps. Some of these disturbances got out of hand and the Earth went into an alternation of extreme ice ages and warm periods several times. During those global ice ages, the oceans became completely covered with sea ice, and the land with large ice sheets. Each time the ice melted on land and sea, you then got a very rapid run-off of nutrients from the released land into the sea. These were times when the sea became so nutrient-rich that a global bloom of photosynthetic cells followed. The production of new cells via photosynthesis (and hence oxygen gas) during that bloom was faster than their breakdown. In other words, oxygen production was faster than oxygen consumption. The dead cells of photosynthetic organisms got buried before other organisms could consume them. Primary production (of cells and of oxygen) went faster than consumption. This is the only way to increase oxygen levels on Earth.
Once the oxygen concentration has risen, it usually stays that way. If primary production declines again – for instance, due to a new ice age – then consumption by respiring organisms will decline along with it, as they will have less food. We therefore see oxygen increases, not decreases, during that period. What we also see during this period is that multicellular algae (already created earlier) start to populate the oceans in abundance. This was an important additional contribution to more oxygen in the oceans. Both the shallow and shallow sea now became oxygen-saturated. Consequently, more oxygen could also become trapped in the atmosphere. We can already see this happening between 800 million and 720 million years ago (800 Ma-720 Ma). After that, the climate went completely out of control. Between 720 MA and 635 MA, we see the most extreme ice ages ever in the history of our planet. We call that period the Cryogenian. Stone layers from that period show that the Earth was covered with large ice sheets, even at the level of the equator. That (almost) completely ice-covered Earth is called “snowball Earth” in English. Again, when these extreme ice ages ended, a warmer climate and a bloom of photosynthetic organisms in the seas followed each time, and thus a significant increase in oxygen.
After the Cryogenian finally (>635 MA), there was enough oxygen for the first animals to emerge. This is truly a tipping point for our planet. This is the start of the great biodiversity we have on our living planet today. Now look back at the graph we started with, at the top of this page. In phase 4, you see a sharp increase in oxygen. It is at this point that we transition to a world populated by animals and plants, on top of the long-established single-celled organisms. It is also said in English that we are entering “the age of the animals”, the age of the animal kingdom, in which we still live today.
Phase 5: the age of the animals
The trend of rising oxygen continues, and the newly emerging animal world experiments worldwide. First come sponges, then jellyfish and first wormids, and the fascinating group of Ediacara which then return to complete extinction. More on this later. Meanwhile, oxygen concentrations continue to rise on Earth. By the end of this period (around 540 Ma), atmospheric oxygen concentration is estimated to be between 4% and 10%. This is enough to cause the animal world to ‘explode’: the Cambrian explosion. By ‘explode’ is meant that in a very short time (around 20 million years, which is geologically super-short) the animal kingdom suddenly changes from ‘first trysts’ to a very extensive range . The evolution of multicellular animals and plants really gets into a phenomenal gear that has only been so spectacular once in Earth’s history. In fact, all the major modern animal groups we know today emerged in a short time back then. Complete with prey and predators, and dozens of new building plans. This has everything to do with the sufficiently high concentration of oxygen, because it is because of this that sufficient energy could be extracted from food, and a truly complex food web became possible.

Another effect of high oxygen concentrations is the formation of ozone (O3), especially at in the stratosphere at 20-30 km altitude. This is not insignificant because this ozone layer absorbs high-energy UV radiation from the sun. In the water of the sea, you were safe from that dangerous UV radiation anyway. But living safely on land is only possible if the most dangerous UV radiation is absorbed in the atmosphere. The formation of the ozone layer made the land a more livable place for the first time. We therefore see that life – until now almost exclusively found in water – slowly but surely began to colonise the land. However, it took several hundred million years before the continents were truly covered with forests and animal life. But when that happened, it was again the cause of a spectacular jump in oxygen. It started mainly in the Devonian (420-360 Ma), and had its real breakthrough in the Carboniferous (360-299 Ma). Look again at the graph of oxygen on Earth at the top of this page. You can again see a sharp rise in oxygen (which afterwards also drops a bit). The Carboniferous was the most oxygen-rich period ever, thanks to the development of the first large forests worldwide. The vegetation also got buried again before the biomass could be consumed. It is these buried plants and trees that constitute our fossil fuel today. Some estimates speak of an oxygen concentration of 30% in the atmosphere. Known from that period are, for example, dragonflies that grew to a metre in size.
Decision
High oxygen concentrations are only possible on our planet thanks to intense ‘primary production’ by photoynthetic organisms, such as cyanobacteria and later plants. Every time primary production exceeds consumption by respiring organisms, you get an increase in oxygen in the oceans and atmosphere. After billions of years and some remarkable jumps, the Earth finally reached a state that allowed unprecedented biodiversity in the sea and on land. The whole system is dynamic, yet stable enough over the long term to allow “the age of the animals” to continue (for now). Oxygen is a determining factor for the living planet.
